RenaPure
Endotoxin-Retentive Cartridge Filter is the final filter in hemodialysis water treatment systems to remove any remaining bacteria, endotoxin and particulate matter from the water.

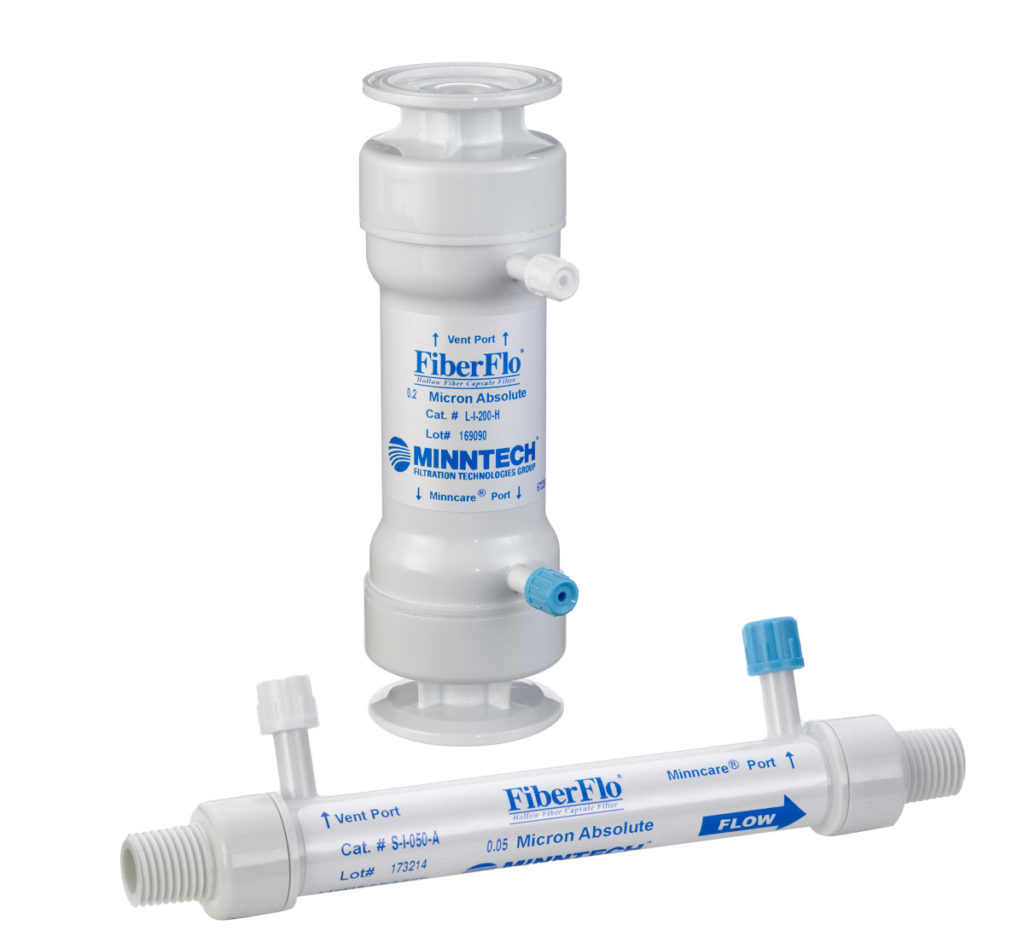
Mar Cor's unique hollow fiber membrane technology is available in FiberFlo brand filtration and separation devices.
Available in either cartridge or capsule form, the filter devices provide fluid purity in high flow applications with minimum filter investment. The unique hollow fiber membrane is made using advanced quality control techniques which produce a remarkably consistent product passing biocompatibility and performance criteria required for registration as a medical device under USFDA regulation 510k.

Hollow Fiber Developer & Collaborator
Mar Cor has the capability to manufacture unique products with private labeling and customized packaging. Being a medical device manufacturer and working under a first rate Quality system makes us a unique partner for development. Our facility is certified to ISO 13485.
From product development to small scale testing to full scale manufacturing, Mar Cor is driven to utilize our internal resources to solve customer filtration requirements.
Development Capabilities
- Over 30 years of OEM and private label manufacturing experience
- Dedicated team of R&D, engineering and quality
- Concept to full scale production
- Vertically integrated manufacturing operation
- Flexibility for production expansion
- Support development and validation/510k submission
- Specialized membranes
- Small scale testing
- Structural integrity / failure analysis
- Wide range of standard module sizes
- Manufactured in the U.S.




